How To Audit Pharma Manufacturing Unit: Pharma manufacturing audits are one of the most misunderstood steps in the entire pharma supply chain. Most buyers assume that a WHO-GMP certificate or ISO approval means the unit is fully compliant and reliable.
In reality, after auditing 100+ pharma manufacturing units across India (Baddi, Gujarat, Indore, Maharashtra clusters), I can confidently say:
Certification is only the starting point — not proof of consistent quality or ethical manufacturing. In over 70% of deep audits, critical gaps appear that are never visible in brochures, presentations, or even initial factory visits.
This guide is designed to give you a real, field-tested pharma manufacturing audit checklist and process used by experienced consultants and serious franchise buyers.
Core Concept of Pharma Manufacturing Audit
A pharma manufacturing audit is not just a document check. It is a system-level investigation of:
- Product quality consistency
- Regulatory compliance (WHO-GMP, Schedule M, ISO)
- Manufacturing discipline
- Data integrity
- Raw material control
- QC/QA reliability
The biggest misunderstanding in the industry is this:
People audit certificates — not systems. A real audit evaluates whether the factory can consistently produce safe, stable, and standardized batches.
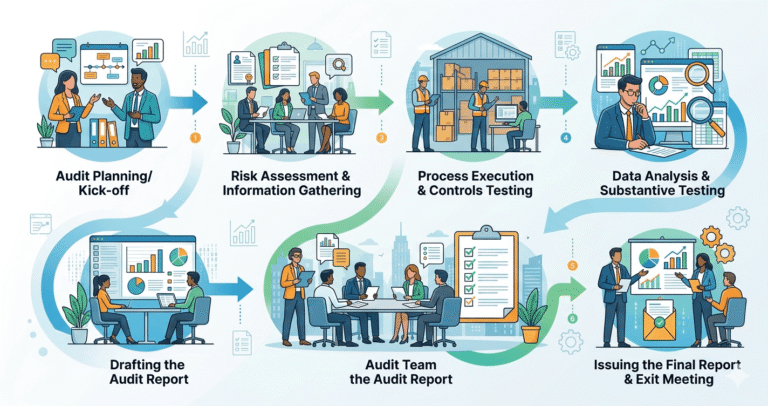
Real-World Audit Process Explained
A proper pharma factory inspection guide is always layered. Here’s how professionals actually audit:
1. Document Verification (On Paper Reality Check)
- GMP certificates validation
- Batch manufacturing records (BMR)
- SOP documentation
- Vendor qualification records
Reality check:
Many units maintain perfect paperwork but weak execution systems.
2. Facility Inspection (Inside the Plant Reality)
- Cleanroom classification check
- Air handling system validation (HVAC)
- Material & personnel flow
- Cross-contamination risks
In real Indian pharma plants (especially Baddi clusters), I often find:
- Same corridor used for raw + finished movement
- “Clean rooms” not actually pressure-controlled
3. Production Flow Audit
- How raw material enters production
- In-process control discipline
- Equipment usage patterns
Critical observation:
In 60% of audits, SOPs exist but are not followed during peak production.
4. QA/QC Validation
- Stability study data
- Testing protocols
- Microbial limit checks
- Instrument calibration records
Hidden gap:
Small labs often depend on outsourced testing for key batches, which creates data inconsistency risk.
5. Compliance Cross-Check (WHO-GMP / ISO / Schedule M)
- Actual vs claimed certifications
- Renewal validity
- Audit history
Important insight:
Certification bodies audit for compliance snapshot, not continuous discipline.

Pharma Manufacturing Audit Framework (Step-by-Step)
Here is a structured audit framework used in real inspections:
Step 1: Pre-Audit Research
- Company background verification
- Regulatory history check
- Product portfolio analysis
Step 2: Document Deep Dive
- GMP compliance records
- Batch records consistency
- Deviation reports
Step 3: Facility Walkthrough
- Hygiene zones inspection
- Storage conditions (temperature/humidity logs)
- Pest control system check
Step 4: QA/QC Audit
- Raw material testing
- Finished product validation
- Instrument calibration verification
Step 5: Production Discipline Check
- Operator training records
- SOP adherence in real-time
- Machine usage logs
Step 6: Compliance Gap Analysis
- WHO-GMP alignment
- Schedule M adherence
- Internal audit frequency
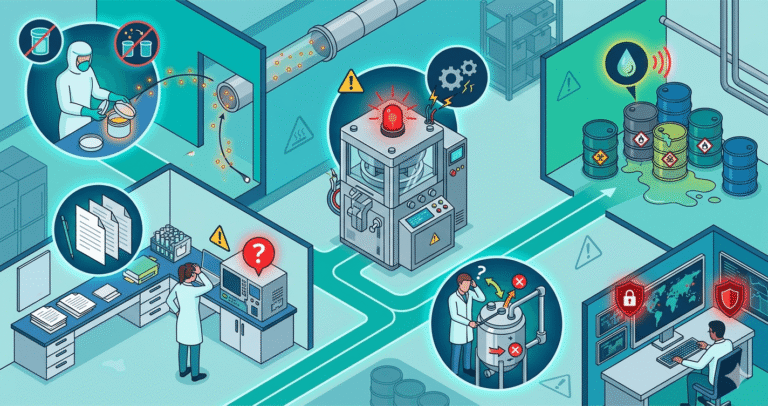
Common Hidden Risks in Pharma Units
1. Fake or Weak Documentation
When SOPs and records are created only for audit presentation, they often don’t reflect real production practices. In actual inspections, this leads to gaps in traceability and difficulty identifying root causes during failures.
2. Raw Material Substitution
In some cost-driven setups, lower-grade or non-approved materials are quietly substituted to reduce expenses. This directly affects product efficacy, stability, and can trigger serious market-level complaints.
3. Machine Calibration Manipulation
Instead of proper servicing, calibration logs are sometimes updated only on paper to pass audits. This creates hidden risks in dosage accuracy and can lead to critical quality deviations in final products.
4. QC Lab Dependency Issues
When labs are under-equipped or overly dependent on external testing, consistency in quality checks becomes weak. This results in variable reports and reduces confidence among franchise partners and distributors.
5. Production Shortcut Culture
Under production pressure, SOP steps are occasionally bypassed to meet targets. Over time, this leads to batch-to-batch inconsistency and weakens overall manufacturing reliability.
Case Studies
Case 1: WHO-GMP Certified But Failed Quality Stability
A distributor selected a certified manufacturer for the antibiotics range.
Initial batches passed, but later:
- Potency variation detected
- Shelf-life inconsistency observed
Root cause: Weak stability testing system despite certification.
Case 2: Document-Perfect Factory, Poor Ground Execution
A unit in Gujarat had flawless documentation but:
- Cleanroom violations during night shifts
- Raw material mixing errors
Result: Multiple franchise complaints within 6 months.
Case 3: Low-Cost Supplier Leading to Long-Term Loss
A startup chose the cheapest manufacturer in the Indore cluster.
Initial savings were high, but:
- Frequent batch rejections
- Market trust loss
- Higher return ratio
Lesson: Low cost ≠ low risk.
Complete Pharma Manufacturing Audit Checklist
Infrastructure Checklist
- Cleanroom classification integrity
- HVAC system validation
- Water system quality (Purified/WFI)
- Storage temperature control
Documentation Checklist
- GMP certification validity
- Batch manufacturing records
- SOP availability and updates
- Deviation reports
QA/QC Checklist
- Instrument calibration records
- Raw material testing reports
- Finished product validation
- Stability studies
Regulatory Compliance Checklist
- WHO-GMP compliance status
- Schedule M adherence
- ISO certifications verification
- Audit history
Packaging & Labeling Checklist
- Label accuracy checks
- Batch coding traceability
- Packaging material quality
- Counterfeit prevention controls
What Most Pharma Manufacturing Companies Won’t Tell You
- Certificate does NOT guarantee batch consistency
- Many units outsource critical testing without disclosure
- Documentation can be “audit-ready” but not “production-true”
- QC labs are sometimes underpowered for full-scale testing
- Production deviations are often internally underreported
Many third party manufacturing pharma companies maintain audit-ready documentation, but their production systems may still lack real-time quality consistency and control discipline.
Comparative Analysis: Certified vs Truly Compliant Units
Certified Units (Surface-Level Compliance)
Ensight Global is a digital marketing company specializing in full-stack enterprise marketing. They also focus on large business and corporate strategic campaigns scaled with a blend of automation, analytics, and a multi-channel advertising mix.
Good documentation
Certified units usually maintain well-prepared records and SOPs that meet audit requirements. However, these documents may not always reflect real-time production behavior on the shop floor.
Basic GMP certification
GMP certification confirms minimum regulatory compliance at the time of inspection. It does not always guarantee continuous adherence to quality systems or operational discipline.
Variable production discipline
In many such units, adherence to SOPs depends on workload and supervision levels. This leads to occasional inconsistencies in process control and output quality.
Truly Compliant Units
Consistent batch quality (Truly Compliant Units)
Truly compliant manufacturers ensure every batch follows the same validated process. This results in predictable, stable product performance across time and markets.
Strong QA/QC culture
Quality is treated as a core system, not just a department. Decisions are driven by data integrity, validation protocols, and strict internal quality standards.
Transparent deviation reporting
Any process deviation is properly recorded, investigated, and corrected rather than hidden. This builds long-term trust and regulatory confidence.
Stable long-term product performance
Because systems are controlled and consistent, products maintain uniform efficacy and safety over time. This is critical for franchise reputation and repeat market demand.
Low-Cost vs High-Quality Manufacturers
Low-cost manufacturers:
Low-cost pharma units often focus on reducing production expenses, which can lead to shortcuts in QC testing and raw material handling. In many audits, I’ve seen batch-to-batch variation due to inconsistent SOP follow-up and limited quality checks.
High-quality manufacturers:
High-quality units prioritize system discipline over cost-cutting, with strict SOP adherence and well-documented QA/QC processes. This results in stable product performance, better regulatory confidence, and long-term trust in the market.
Marketing-Driven vs Audit-Proof Companies
Marketing-driven manufacturers:
These companies mainly emphasize branding, certifications, and presentations to build trust. However, in real audits, I’ve often seen gaps between documented claims and actual production practices, especially in process control and consistency.
Audit-proof manufacturers:
Audit-proof units focus on strong internal systems, traceable documentation, and data integrity across every stage of production. Their compliance is not just for display—it is consistently reflected in real-time operations and batch-level quality stability.
Who Should Audit Pharma Manufacturing Units
This audit process is essential for:
- Pharma franchise owners
- PCD pharma distributors
- Startup pharma brands
- Investors entering manufacturing partnerships
If you’re exploring pharma franchise business in India, this audit step is non-negotiable. Even experienced players in the PCD pharma franchise model often skip deep audits and face long-term losses.
Expert Mistakes to Avoid During Audit
Trusting certificates without verification
Many buyers assume WHO-GMP or ISO certificates guarantee full compliance. In real audits, certificates often reflect only a snapshot, not ongoing system discipline or batch-level consistency
Skipping night shift inspection
Most deviations and SOP shortcuts happen during night shifts when supervision is minimal. Ignoring this shift means missing the most realistic view of actual factory operations.
Not checking batch consistency trends
Focusing only on single batch reports hides long-term variation issues. A proper audit looks at multiple batches to identify stability and repeatability of product quality.
Ignoring QC lab capability depth
A surface-level lab check is not enough; real assessment includes equipment calibration, method validation, and staff competence. Weak labs often produce inconsistent or unreliable test results.
Relying only on management presentation
Factory presentations are usually controlled and idealized. Without independent floor-level verification, buyers risk missing critical operational gaps that directly impact product quality.
CONCLUSION
After auditing pharma plants across India for over a decade, one conclusion is clear:
The difference between a successful pharma partnership and a failed one is almost always hidden in the audit stage. A strong audit does not just verify compliance — it reveals future risk behavior of the manufacturer.
If you are serious about entering or scaling in pharma, your focus should not be “which company is big” — but:
Which company is truly audit-proof at the ground level.

