Tablet Manufacturing Process In Pharma Industry looks simple on paper—mix, compress, pack. But inside real pharma plants, it’s one of the most sensitive, failure-prone processes.
A small mistake in moisture, blending, or compression can lead to batch rejection, market complaints, or complete product failure.
If you’re a pharma student, a beginner, or planning a PCD pharma franchise in India, understanding this process is not optional—it directly impacts your product quality, brand reputation, and profitability.
In this guide, you’ll learn:
- Step-by-step tablet manufacturing process
- What actually happens inside production units
- Common problems (and why they occur)
- Real-world business impact
- How to choose the right manufacturer
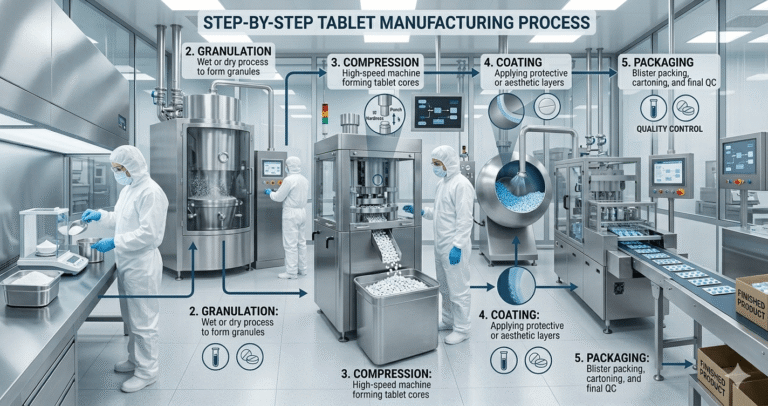
Step-by-Step Tablet Manufacturing Process In Pharma Industry
1. Raw Material Selection & Testing
This is the foundation of the entire tablet manufacturing process, as every step that follows depends on the quality and consistency of raw materials. Any compromise at this stage directly impacts formulation performance, stability, and final product reliability.
Why it matters:
- API (Active Pharmaceutical Ingredient) defines therapeutic effect
- Excipients control flow, compression, and stability
What happens in real plants:
- Raw materials are tested for:
- Identity
- Purity
- Particle size
- Moisture content
Real issue:
In 40–50% of small-scale units, expedient quality is compromised to reduce cost.
Impact:
- Poor flow → weight variation
- Impure API → therapeutic failure
2. Weighing & Dispensing
Precise weighing of each ingredient is critical to ensure every tablet contains the correct dosage. Even a small deviation at this stage can lead to content uniformity issues, affecting both product quality and patient safety.
Why it matters:
- Even a slight deviation affects dosage uniformity
Common mistake:
- Manual weighing errors in non-automated plants
Real insight:
Many small manufacturers skip double verification to save time, leading to batch inconsistencies.
3. Mixing / Blending
Uniform mixing of API and excipients ensures that every tablet delivers the same dose and therapeutic effect. Without proper blending, some tablets may contain too much or too little drug, leading to serious quality and compliance issues.
Why it matters:
- Ensures every tablet has the same dose
What can go wrong:
- Under-mixing → dose variation
- Over-mixing → segregation
Real plant issue:
In 60% of cases, improper blending time causes content uniformity failures.
Impact:
- Regulatory rejection
- Doctor distrust
4. Granulation (Wet & Dry)
Granulation is one of the most critical stages in the Tablet Manufacturing Process In Pharma Industry, as it directly affects flow and compression.
Wet Granulation
- Uses binder solution
- Improves compressibility
Problems:
- Over-wetting → sticky granules
- Under-wetting → poor binding
In real plants, moisture control is one of the biggest challenges.
Dry Granulation
- No liquid used
- Suitable for moisture-sensitive drugs
Problems:
- Poor compaction → weak tablets
5. Drying
This step removes excess moisture from granules to ensure proper compression and stability. Controlled drying is essential, as both excess and insufficient moisture can negatively affect tablet strength and shelf life.
Why it matters:
- Moisture directly affects tablet hardness and stability
Common issue:
- Uneven drying
In 60–70% of manufacturing units, granulation issues arise due to improper drying temperature control.
Impact:
- Too dry → brittle tablets
- Too wet → sticking during compression
6. Milling
Granules are sized uniformly to ensure smooth flow and consistent die filling during compression. Uniform size distribution helps maintain tablet weight, hardness, and overall quality.
Why it matters:
- Uniform size = better flow + compression
Real issue:
- Over-milling → fine powder → poor flow
- Under-milling → uneven tablets
7. Lubrication
Lubrication is a small step but highly sensitive—insufficient lubricant causes sticking, while excess lubrication can weaken tablets. In real manufacturing, over-lubrication is a common mistake that affects dissolution performance.
Why it matters:
- Prevents sticking to punches
Critical mistake:
- Over-lubrication
Many manufacturers overuse lubricant to avoid machine sticking.
Impact:
- Poor dissolution
- Reduced drug release
8. Tablet Compression
Compression is the final and most sensitive stage of the Tablet Manufacturing Process In Pharma Industry, where defects are most likely to occur.
Machines used:
- Rotary compression machines
Common problems:
| Problem | Cause | Impact |
|---|---|---|
| Capping | Air entrapment | Tablet breaks |
| Lamination | Improper compression | Layer separation |
| Weight Variation | Poor flow | Dose inconsistency |
| Hardness Issues | Pressure variation | Dissolution failure |
Note: In real plants, compression-related defects are among the most frequent batch rejection causes.
9. Coating (If Applicable)
Types:
- Film coating
- Sugar coating
- Enteric coating
Why it matters:
- Taste masking
- Controlled release
- Aesthetic appeal
Real issue:
Many small manufacturers compromise on coating quality to reduce cost.
Problems:
- Peeling
- Color variation
- Poor finish
Business impact:
- Poor appearance = low doctor trust
10. Quality Control & Packaging
Final testing includes:
- Weight variation
- Hardness
- Friability
- Dissolution
- Stability
Real issue:
Some third-party manufacturers rely on minimal QC to save cost.
Impact:
- Market complaints
- Product returns
Wet Granulation vs Dry Granulation vs Direct Compression
| Parameter | Wet Granulation | Dry Granulation | Direct Compression |
|---|---|---|---|
| Cost | High | Medium | Low |
| Complexity | High | Medium | Low |
| Risk | Moisture issues | Compaction issues | Segregation risk |
| Best For | Most formulations | Moisture-sensitive drugs | Simple formulations |
Reality Check: Many manufacturers prefer direct compression to reduce cost, even when it’s not ideal.
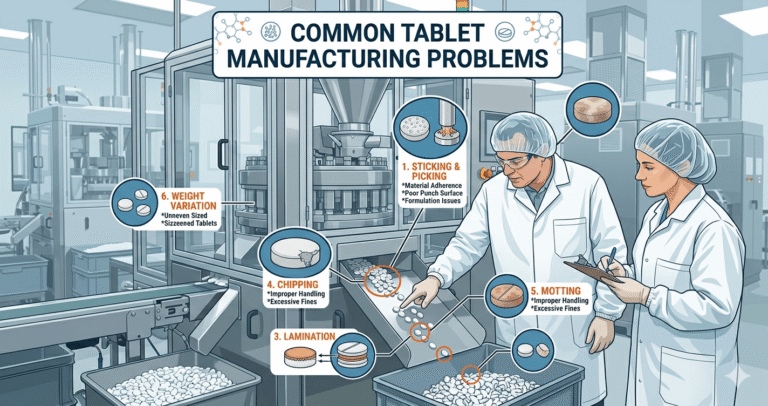
Common Problems in Tablet Manufacturing Process In Pharma Industry
1. Capping
Capping happens when air gets trapped inside the tablet during compression, causing the top or bottom layer to separate. In real manufacturing, this often occurs due to low moisture or high machine speed. If not corrected, it leads to fragile tablets and high batch rejection.
2. Lamination
Lamination is when a tablet splits into multiple layers, usually due to excessive compression force or improper formulation balance. In practical scenarios, it reflects poor process control and can make tablets unstable during transport and storage.
3. Weight Variation
Weight variation occurs when powder does not flow uniformly into the die cavity, leading to uneven tablet weights. This is a common issue in plants with poor granulation or blending, and it directly affects dosage accuracy and regulatory compliance.
4. Hardness Issues
Hardness problems arise when tablets are either too soft or too hard due to incorrect binder levels or compression pressure. In real cases, this imbalance can cause tablets to break easily or fail dissolution, impacting both quality and patient effectiveness.
5. Dissolution Failure
Dissolution failure means the tablet does not release the drug properly in the body, often due to over-lubrication or poor coating. This is one of the most serious issues, as it directly affects therapeutic performance and can lead to product rejection in the market.

What Most Pharma Manufacturers Won’t Tell You
This is where the gap between documented processes and actual factory practices becomes clear. What looks perfect on paper often changes when production pressure, cost constraints, and timelines come into play.
Cost Cutting in Excipients
To improve margins, some manufacturers use lower-grade fillers or substitute excipients without proper evaluation. In real production, this directly affects tablet stability, dissolution, and overall consistency—something that often shows up only after the product reaches the market.
Skipping Validation Batches
Validation is meant to ensure that every batch performs the same way. However, in many small or cost-driven units, this step is rushed or partially skipped to save time and money. The result is unpredictable batch quality and higher chances of failure during scale-up.
Overloading Machines for Higher Output
To maximize daily production, machines are often run beyond optimal capacity. While this increases short-term output, it leads to compression issues like capping, weight variation, and faster equipment wear—ultimately increasing long-term losses.
Outsourcing Risks in Third-Party Manufacturing
In third party pharma manufacturing, consistency is one of the biggest hidden risks. Different batches may be produced at different times or even different facilities, leading to variation in quality, finish, and performance. In many cases, what you see in samples is not what you get in bulk production.

Real Case Scenarios
Case 1: Moisture Imbalance Failure
A batch failed dissolution due to excess moisture in granules.
Result:
- Entire batch rejected
- Loss: ₹3–5 lakh
Case 2: Coating Complaint
A PCD company received complaints of peeling tablets.
Reason:
- Poor coating quality
Impact:
- Distributor stopped orders
Case 3: Weight Variation Issue
Improper blending caused uneven API distribution.
Outcome:
- Regulatory warning
- Brand damage

Business Impact of Poor Tablet Manufacturing
If you are running a pharma franchise business model, manufacturing quality directly affects:
- Distributor trust
- Doctor prescription rate
- Repeat orders
- Brand positioning
Example:
- Poor dissolution → doctor stops prescribing
- Bad coating → low market acceptance
- Batch failure → financial loss
Poor execution of the Tablet Manufacturing Process In Pharma Industry can lead to product returns, doctor rejection, and brand damage.
How to Choose a Good Tablet Manufacturer (5-Step Checklist)
Step 1: Check WHO-GMP Certification
WHO-GMP certification shows that the manufacturer follows standardized quality and safety practices. However, don’t rely on the certificate alone—many issues still happen on the shop floor. Always cross-check how strictly these standards are actually followed in daily production.
Step 2: Ask for Batch Records
Batch records give you a clear picture of how consistently products are manufactured. In real scenarios, variations in mixing time, drying, or compression often show up here. Reviewing these records helps you identify hidden inconsistencies before they affect your business.
Step 3: Evaluate QC Reports
Quality Control reports reveal whether the product passes key tests like dissolution, hardness, and stability. A good manufacturer will have detailed and consistent reports across batches. Inconsistent or incomplete data is often a red flag for future product complaints.
Step 4: Check Product History
Always check if the manufacturer has faced past market complaints, returns, or quality issues. In real pharma business, past performance is one of the strongest indicators of future reliability—especially in competitive markets.
Step 5: Visit the Plant
A physical visit tells you what documents can’t—actual hygiene, machine condition, staff discipline, and process control. Many problems like overloading machines or poor handling practices are only visible when you see the plant in operation.
Connection With Pharma Business
Whether you’re:
- Starting a pharma franchise
- Entering third party pharma manufacturing
- Scaling a product portfolio
Your success depends on manufacturing quality. Most failures in starting a pharma franchise happen not due to marketing—but due to poor product performance.
CONCLUSION
Understanding the Tablet Manufacturing Process In Pharma Industry gives you a major advantage in ensuring product quality and long-term business success.
The difference between a successful pharma brand and a struggling one often comes down to:
- Process control
- Manufacturer selection
- Quality consistency
If you understand what happens inside the plant, you gain a massive advantage over competitors who rely blindly on third-party manufacturers. And in pharma, quality is not optional—it’s survival.

